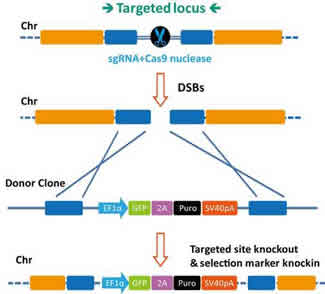
This approach is particularly useful for genotyping when a reference genome is not available, such as in ecological studies (Andrews et al., 2016). RAD-seq is a protocol for genotyping and discovery of single-nucleotide polymorphisms (SNPs) (Baird et al., 2008). It should be checked that the gene of interest is in frame with an eventual C-terminal tag.Restriction-Site Associated DNA Sequencing The overlap between the primer and the strand complement to the 3'-end of the gene of interest should be long enough to give a T m of 60☌ or more (calculated as shown above). Overlap with the stand complement to the 3'-end of the gene of interest.To increase the efficiency of termination it is possible to use 2 or 3 stop codons in series. A stop codon ( TAA is preferred because it is less prone to read-through than TAG and TGA) should be included when no C-terminal tag is used. 210-211 of the 2000/01 catalog of New England Biolabs.
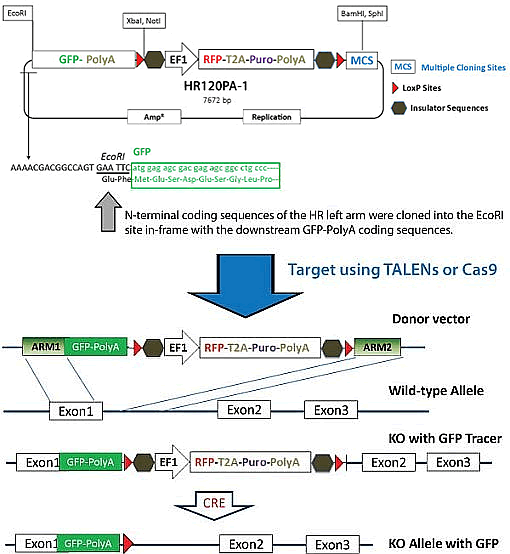
Data on the effect of the extension length and sequence on the cleavage efficiencies of the most used restriction enzymes can be found on pp. A 5' extension of the restriction site with 2-10 bases greatly increases the cleavage efficiency of most enzymes. Restriction enzymes cleave DNA much less efficient towards the end of a fragment. Alternatively, you could pick any restriction enzyme that gives a blunt end upon cleavage (see cloning) The restriction site should be the same or provide the same sticky end to the second of the restriction enzymes in the multiple cloning site of the vector chosen to clone the gene of interest into. The 3'-end primer overlaps with the DNA strand complementory to the 3'-end of the gene of interest and should contain the following elements: The overlap between the primer and the gene of interest should be long enough to give a T m of 60☌ or more (calculated as shown above). It should be checked that the start codon and the gene of interest are in frame with an eventual N-terminal tag and/or fusion partner. A start codon (usually ATG) should be included when the gene of interest is not expressed with an N-terminal tag or fusion partner or when an N-terminal methionine residue is present. Data on the effect of the extension length and sequence on the cleavage efficiencies of the most used restriction enzymes can be found in the reference appendix of the New England Biolabs catalogue.

Often Nco ICC ATGG) or Nde I (CAT ATG) are chosen because the ATG within these sites can be used directly to create the ATG start codon and/or the ATG codon for the N-terminal methionine residue (see Utilisation of the Nco I cloning site ) Alternatively, you could pick any restriction enzyme that gives a blunt end upon cleavage (see cloning). The restriction site should be the same or provide the same sticky end to the first of the restriction enzymes in the multiple cloning site of the vector chosen to clone the gene of interest into. The 5'-end primer overlaps with the 5'-end of the gene of interest and should contain the following elements: The GC content of a primer should be between 40 and 60%. The T m for a primer can be estimated using the following formula: T m = 2☌ * (A + T) + 4☌ * (C + G) Usually good results are obtained when the T m's for both primers are similar (within 2-4 ☌) and above 60☌. The specificity of PCR depends strongly on the melting temperature (T m) of the primers (the temperature at which half of the primer has annealed to the template). Shorter primers could lead to amplification of nonspecific PCR products. Usually a primer length of 18-30 bases is optimal for most PCR applications. Therefore, it is possible to add sequence elements, like restriction sites, to the 5'-end of the primer molecule. This often leads to primer-dimer formation.īases at the 5'-end of the primer are less critical for primer annealing. Primer pairs should be checked for complementarity at the 3'-end. a 3' thymidine, since it is more prone to mispriming than the other nucleotides.This may stabilize nonspecific annealing of the primer. 3 of more G or C bases at this position.Especially the 3'-end of the primer molecule is critical for the specificity and sensitivity of PCR. The first step is the design of the necessary primers. The gene of interest usually has to be amplified from genomic or vector DNA by PCR (polymerase chain reaction) before it can be cloned into an expression vector. Transformation of competent E.coli cells.EMBL Technology Developers Programme (ARISE).EMBL International Centre for Advanced Training (EICAT).
